608
The first direct evidence of a stable carbine - the most durable material in the world
Theoretically, the carbine is 40 times stronger than diamond
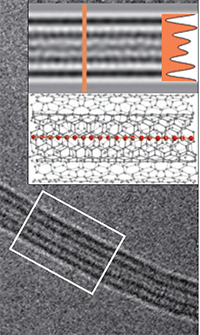
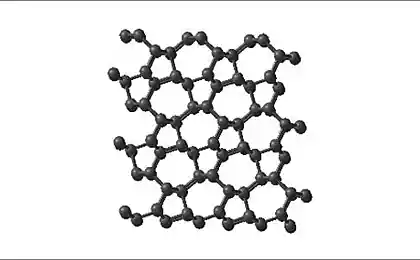
Carbon is very diverse in its modifications. There are many carbon allotropes, which have unique properties: diamond, graphite, fullerene, etc., only about a dozen allotropes. But in all this diversity was one exception - carbine. This allotropic form of carbon consists of fragments with a triple bond -S≡S-.
Carbine could not manage to synthesize, but the properties of the material studied for a long time. The reason for failure is that the carbine extremely unstable.
Failing for the failure, the researchers speculated that some of the mechanical properties of the carbine must exceed the properties of all known carbon allotropes. It was assumed that its mechanical stiffness of 2-fold greater than that of graphite; strength - 40 times more than that of diamond; the tensile strength is also greater than any form of carbon. Well, and other scientists believed that the stable form carbine does not exist.
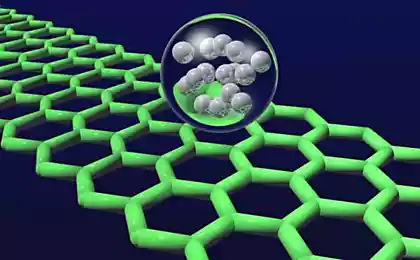
Now in this dispute come to an end. Extra long-1D molecule carbine we see right in the photo (on the center of the nanotube).
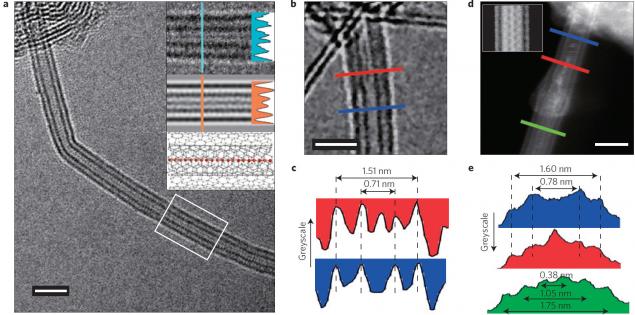
An international group of scientists for the first time found a way to mass production of stable carbine. By "mass production" means the compilation of long chains of atoms that are stable.
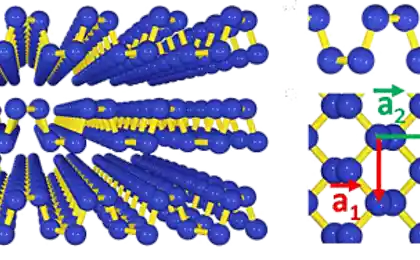
For the production of the material took two layers of graphene, we squeezed them together and tied in thin carbon nanotubes with a double wall. These nanotubes are surrounded by 1D-molecule carbine and protect it from imminent disintegration.
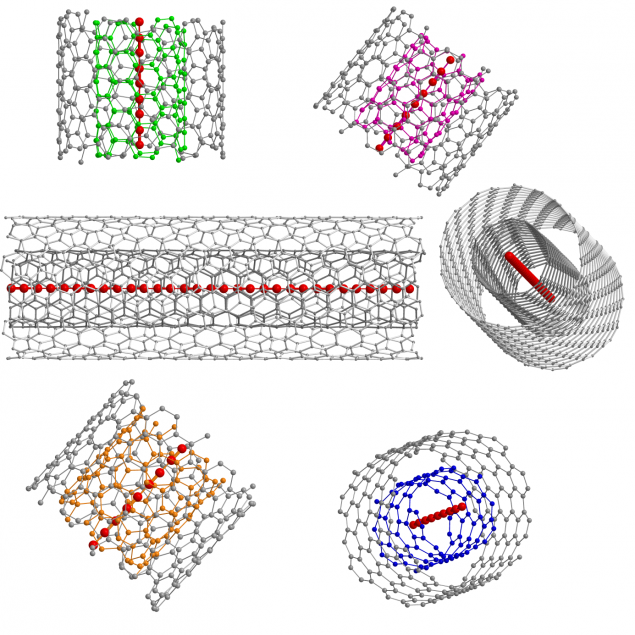
Until today, the maximum number of carbon atoms in a continuous chain was 100 (2003 year). Now the record is beaten, and very important: a new method allowed to make a chain of atoms 6400, and this is not the limit
. In addition, improved electrical properties of the carbine with a prolongation of the chain. This means that scientists will create material for interesting experiments.
The article "Closed linear carbon chains as a way to mass production carbine" published in the journal Nature Materials (doi: 10.1038 / nmat4617; pdf).
It's nice that in the published article, there are several references to the work of Russian scientists, including an article in the journal "Nature" of 1969, where Soviet physicists sweet and Kudryavtsev describes the properties of diamond, graphite and carbine as the allotropic forms of carbon.
< br> It should be noted that the Nobel Prize in Physics has recently awarded for his fundamental work on other allotropic forms of carbon: fullerene (1996) and graphene (2010), so that the synthesis carbine - a valuable task for scientists.
Source: geektimes.ru/post/274350/

Carbon is very diverse in its modifications. There are many carbon allotropes, which have unique properties: diamond, graphite, fullerene, etc., only about a dozen allotropes. But in all this diversity was one exception - carbine. This allotropic form of carbon consists of fragments with a triple bond -S≡S-.
Carbine could not manage to synthesize, but the properties of the material studied for a long time. The reason for failure is that the carbine extremely unstable.
Failing for the failure, the researchers speculated that some of the mechanical properties of the carbine must exceed the properties of all known carbon allotropes. It was assumed that its mechanical stiffness of 2-fold greater than that of graphite; strength - 40 times more than that of diamond; the tensile strength is also greater than any form of carbon. Well, and other scientists believed that the stable form carbine does not exist.
Now in this dispute come to an end. Extra long-1D molecule carbine we see right in the photo (on the center of the nanotube).

An international group of scientists for the first time found a way to mass production of stable carbine. By "mass production" means the compilation of long chains of atoms that are stable.
For the production of the material took two layers of graphene, we squeezed them together and tied in thin carbon nanotubes with a double wall. These nanotubes are surrounded by 1D-molecule carbine and protect it from imminent disintegration.

Until today, the maximum number of carbon atoms in a continuous chain was 100 (2003 year). Now the record is beaten, and very important: a new method allowed to make a chain of atoms 6400, and this is not the limit
. In addition, improved electrical properties of the carbine with a prolongation of the chain. This means that scientists will create material for interesting experiments.
The article "Closed linear carbon chains as a way to mass production carbine" published in the journal Nature Materials (doi: 10.1038 / nmat4617; pdf).
It's nice that in the published article, there are several references to the work of Russian scientists, including an article in the journal "Nature" of 1969, where Soviet physicists sweet and Kudryavtsev describes the properties of diamond, graphite and carbine as the allotropic forms of carbon.
< br> It should be noted that the Nobel Prize in Physics has recently awarded for his fundamental work on other allotropic forms of carbon: fullerene (1996) and graphene (2010), so that the synthesis carbine - a valuable task for scientists.
Source: geektimes.ru/post/274350/
St. Petersburg photographer commuters compared to their profiles "VKontakte"
How to quickly learn a foreign language, if you're over 30