467
Nissan is working on solid oxide fuel cell
On the way to creating electric vehicles with long range of travel, Nissan is developing a solid oxide fuel cell that directly converts ethanol to electricity for powering electric cars.
While the range of modern electric cars hovering around 500 km, Nissan claims that its system of solid oxide fuel cells can offer a ride, a range over 600 km. This puts him in the same line with most gasoline cars, but with the same quiet running, confident acceleration and linear gas pedal as on electric vehicles.
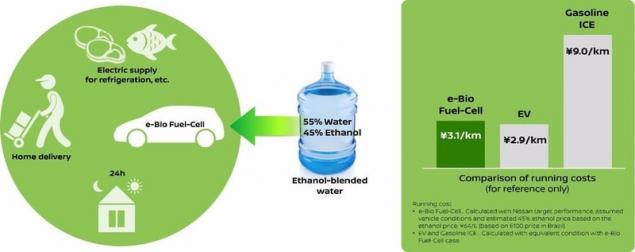
According to Nissan, the car has a fuel cell tucked up to 55 percent water and 45 percent with a mixture of ethanol, operating costs only slightly higher than they would when driving a normal electric vehicle, and you will pay about two thirds less than using a petrol vehicle.
This system also supports the existing infrastructure of gas stations, and does not require special tanks with liquid fuel and a unique valve like most cars with hydrogen fuel cells, or charging station, in the case of electric vehicles.
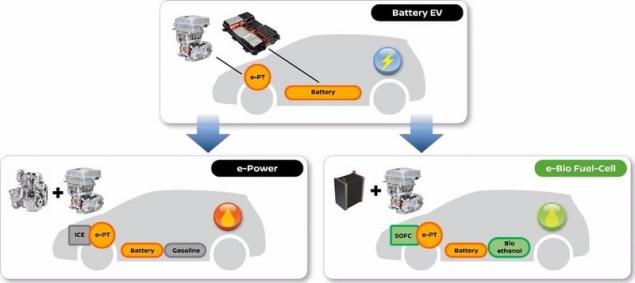
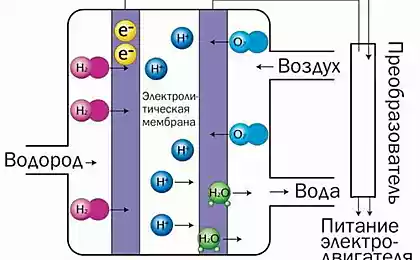
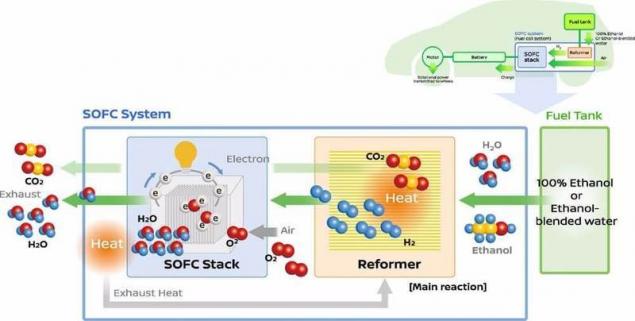
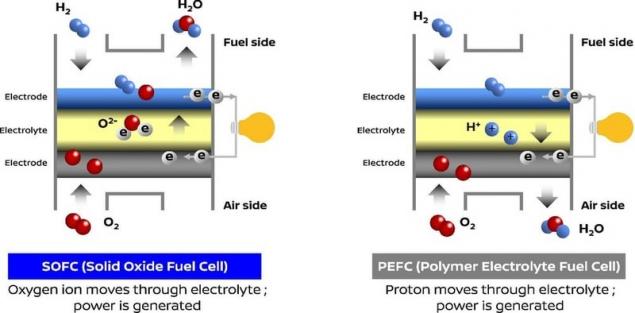
So, how does it work? The fuel cell uses a reformer to convert bio-ethanol to hydrogen. This hydrogen is then mixed with atmospheric air, and is converted into electrical energy using a fuel cell, providing the battery supplies power to the motor.
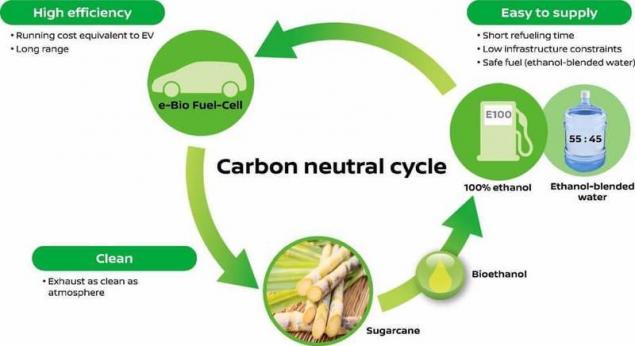
Nissan says that the cycle of using ethanol to power a car, it is carbon-neutral, CO2 emissions neytralizuya in the process of growing sugarcane, which is bio-fuel.
This theory of course ignores the fossil fuel burning that is involved in the processing and transport of biofuels, not to mention the other problems associated with biofuel production based on agricultural crops, but that's another story.
At the moment this technology is still under development. It uses an amorphous-silicon battery as a potential way for Nissan to improve driving range and relevance of electric vehicles.
P. S. And remember, only by changing their consumption — together we change the world! ©
Source: facepla.net/the-news/tech-news-mnu/5486-nissan-%D1%82%D0%BE%D0%BF%D0%BB%D0%B8%D0%B2%D0%BD%D1%8B%D0%B9-%D1%8D%D0%BB%D0%B5%D0%BC%D0%B5%D0%BD%D1%82.html
While the range of modern electric cars hovering around 500 km, Nissan claims that its system of solid oxide fuel cells can offer a ride, a range over 600 km. This puts him in the same line with most gasoline cars, but with the same quiet running, confident acceleration and linear gas pedal as on electric vehicles.

According to Nissan, the car has a fuel cell tucked up to 55 percent water and 45 percent with a mixture of ethanol, operating costs only slightly higher than they would when driving a normal electric vehicle, and you will pay about two thirds less than using a petrol vehicle.

This system also supports the existing infrastructure of gas stations, and does not require special tanks with liquid fuel and a unique valve like most cars with hydrogen fuel cells, or charging station, in the case of electric vehicles.

So, how does it work? The fuel cell uses a reformer to convert bio-ethanol to hydrogen. This hydrogen is then mixed with atmospheric air, and is converted into electrical energy using a fuel cell, providing the battery supplies power to the motor.

Nissan says that the cycle of using ethanol to power a car, it is carbon-neutral, CO2 emissions neytralizuya in the process of growing sugarcane, which is bio-fuel.

This theory of course ignores the fossil fuel burning that is involved in the processing and transport of biofuels, not to mention the other problems associated with biofuel production based on agricultural crops, but that's another story.
At the moment this technology is still under development. It uses an amorphous-silicon battery as a potential way for Nissan to improve driving range and relevance of electric vehicles.
P. S. And remember, only by changing their consumption — together we change the world! ©
Source: facepla.net/the-news/tech-news-mnu/5486-nissan-%D1%82%D0%BE%D0%BF%D0%BB%D0%B8%D0%B2%D0%BD%D1%8B%D0%B9-%D1%8D%D0%BB%D0%B5%D0%BC%D0%B5%D0%BD%D1%82.html
Geothermal power plant at Menengai 400 MW in Kenya will start in July this year
Happiness love peace