2163
Hollow iron oxide nanoparticles as the future of the electrodes of lithium-ion batteries
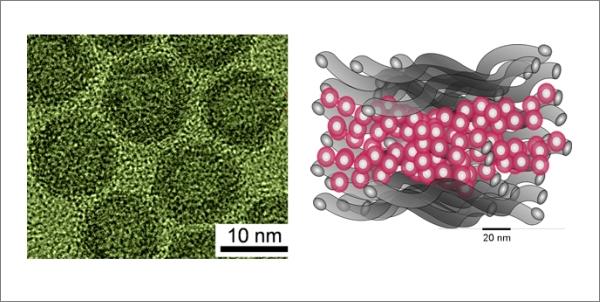
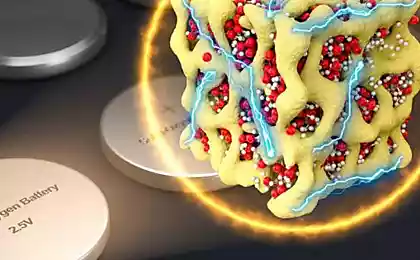
Left - photomicrograph of hollow nanoparticles of iron oxide; Right - a schematic representation of the innovation-based cathode hollow nanoparticles between the layers of carbon nanotubes (Figure Argonne National Laboratory).
Specialists from the Argonne National Laboratory and the University of Chicago (USA) synthesized hollow iron oxide nanoparticles with a high concentration of point defects to implement a completely new concept of production of electrodes based on nanoparticles sandwiched between two layers of pure carbon nanotubes.
When a new electrode becomes the cathode, high vacancy concentration of iron in the nanoparticles can significantly improve the performance of existing lithium-ion batteries, say the developers.
Conventional electrodes based on nanoparticles pretty quickly lose efficiency due to poor contact between the nanoparticles and collectors. New same electrodes allow reversible intercalation of lithium ions, which results in a high capacity and efficiency, superior performance and excellent charge stability (performance constant for more than 500 charge cycles). Progress - a clear proof that the morphology of the nanomaterial cathode is the most critical factor for the further development of lithium-ion batteries.
Hollow nanoparticles gamma-Fe2O3 were synthesized at Argonne National Laboratory with the amount of iron vacancies four times superior to those of conventional nanoparticles. An innovative method of production of the electrode is locked layer of nanoparticles between two layers of net multiwall carbon nanotubes without using "link" or other additives.
Electrochemical tests showed a high capacity (132 mA ∙ h / g at 2, 5), the highest faradic efficiency is 99, 7% (demonstrates the effectiveness of the electron transport chain in the electrochemical), excellent high-speed characteristics (133 mA ∙ h / g at 3 000 mA / g) and solid stability (as already said, more than 500 cycles). In addition, in the present interesting data about the internal structural transformation of nanoparticles obtained by synchrotron X-ray absorption and diffraction, which give a clear idea of what is going on with lithium during electrochemical cycle.
Report on the work can be found in the journal Nano Letters.
Detect dark matter particles XENON100 detector has not yet been
The half-century mystery intermediate carbon structure solved: this M-carbon