540
Harmful fast charger for battery

A new study has shown that fast charging a Li-ion battery, and then it very fast discharge may not be as destructive as previously thought.
And now that the advantages of slow charging and discharging was overrated. The study was conducted by researchers from Stanford University and the Institute of Materials science And Energy Stanford (Stanford Institute for Materials & Energy Sciences — SIMES) at the Department of Energy National Laboratory Accelerators (DOE at SLAC National Accelerator Laboratory, which is developing particle accelerators), with the assistance of colleagues from Sandia National Laboratory (Sandia National Laboratories), the American branch of the Institute of Technology Samsung (Samsung Advanced Institute of Technology America) and the National Laboratory Lawrence Berkeley (Lawrence Berkeley National Laboratory).
The results refute the prevailing view that ultra-fast charging batteries has a negative impact on the condition of the electrodes compared to the charge at a slower speed. Besides, probably, scientists will be able to modify electrodes or a method of charging batteries to ensure more uniform discharge process and charge the batteries and extend their service life.
"The processes occurring at the electrodes during charging and discharging are among the many factors that determine battery life, but until this study, these processes have not been thoroughly studied. We moved to a new level of understanding of the degradation of the battery characteristics," says the study's lead author William Chu (William Chueh) from SIMES.
According to him, the research results can be applied directly to modify currently installed in lithium-ion batteries oxide and graphite electrodes. And at least half of those that are currently in development.
A very important source of battery wear are the swelling and shrinkage of the negative and positive electrodes as they absorb and release ions of the electrolyte during charging and discharging.

During the study, the researchers conducted monitoring lithium-iron-phosphate cathode. If most or all of the nanoparticle material actively involved in the process of zaradki and discharge, they absorb and release ions more gently and uniformly. However, if only a small percentage of particles of the material involved in the process, they have a tendency to rapid decay, and destruction, reducing the battery performance.
Previous studies have raised contrary views on the behavior of the nanoparticles of the cathode material. To dig deeper, scientists have created a small, coin-size batteries, loaded them using a different combination of voltage and duration of the charging process, and then quickly disassembled and washed components to stop the process of charging/discharging. Then they cut the electrode into extremely thin layers, and Berkeley lab has subjected them to intense x-ray synchrotron radiation at the Advanced Light Source located at the center for applied research one of the branches of DOE.
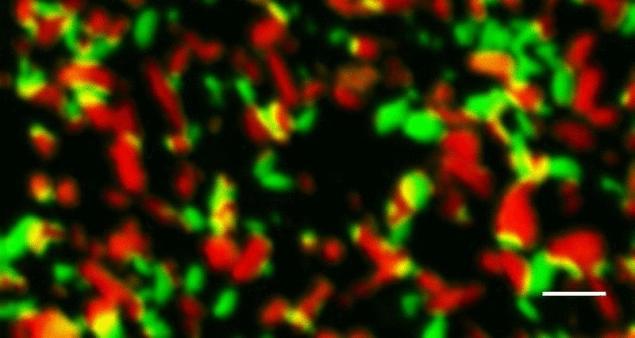
The made x-ray microscope and it displayed nanoparticles in the middle of charging process. The color of the particle corresponds to the charge level: fully charged – green, charging – yellow, discharged completely red. Length scale of 500 nm. The photo was taken at the SLAC National Accelerator Laboratory.
"We were able to see thousands of electrode nanoparticles at a time and take pictures of them at different stages of the processes of charging and discharging. This study is the first sequential work carried out with the use of completely different stages of the processes of charging and discharging," says one of the leading researchers Jiang Lee (Li Yiyang).
Analyzing the data using the model developed in Massachussets Institute of Technology, researchers found that only a small part of nanoparticles absorbs and releases ions in the discharge process, even when it makes at high speed. However, if the charging rate exceeds some certain value, more and more particles become involved in the process, becoming more uniform and less damaging mode. This suggests that scientists will be able to improve the electrode material or the process for achieving high-speed charging and discharging while maintaining long battery life.
Of course, that the lab results and only one type of batteries, however, they are very encouraging and show that not everything is as bad as it seemed.
At the moment it seems that the change of batteries and will not become mainstream for owners of electric vehicles, despite the fact that Tesla has provided such an option on their cars. But fast charging can be a very important element for long-distance travel. In most cases, electric vehicles will charge at night and will not require any additional charge, but in cases when you need to travel further than allowed by the capacity of the car battery, have a battery that can take a lot of power for a short period of time and without any damage, can become very important.
The next step of the study, according to Lee, will be passing the electrodes through thousands of cycles of charging and discharging in a row to mimic everyday use. Scientists also hope to get pictures of the battery directly in the process without stopping it and without disassembling the battery apart.
Source: facepla.net